Patient-Derived Organoids Reveal the Efficacy of DCLK1-Targeted Kinase Inhibitor DCLK1-IN-1 Against Gastric Adenocarcinoma Stemness and Tumorigenesis
Authors
Jinsen Shi1, Dongfeng Qu2, Yuping Yang1, Zhiyun Cao1, Courtney W. Houchen2, and Nathaniel Weygant1
Affiliations
- Academy of Integrative Medicine, Fujian University of Traditional Chinese Medicine, Fuzhou, CN
- Dept. of Medicine, The University of Oklahoma Health Sciences Center, Oklahoma City, OK, USA
Introduction
Gastric Cancer (GC) is characterized by late diagnosis and cancer stem cell (CSC)-related tumor heterogeneity. Doublecortin-like kinase 1 (DCLK1) is a critical gastrointestinal CSC marker confirmed by self-renewal assays, pathological analysis, and in vivo lineage tracing. Here, we assessed the effect of DCLK1-targeted therapy using GC cell lines and patient-derived organoids (PDOs).
Methods
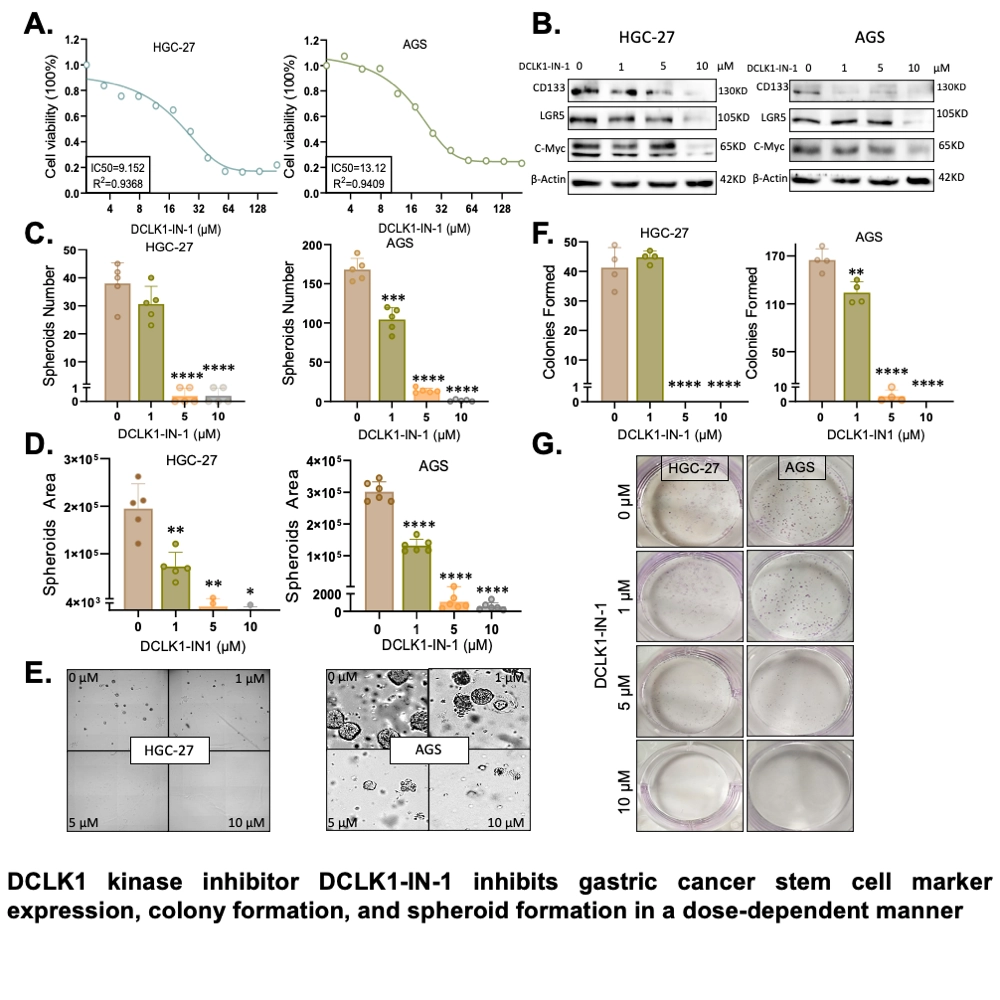
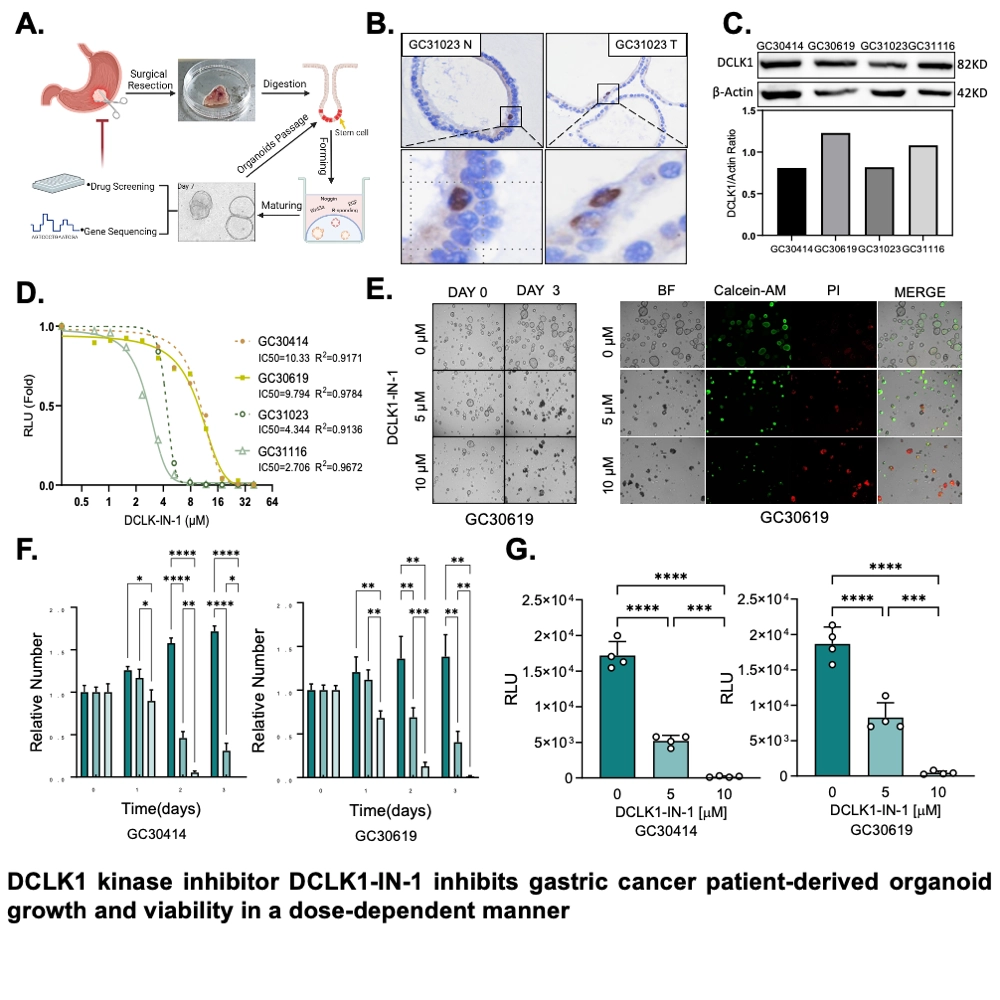
GC cell lines, HGC-27 and AGS were used to perform MTT, colony formation, and spheroid assay after treatment with 0 – 10 μM of DCLK1 kinase inhibitor DCLK1-IN-1. For PDO establishment, GC patient tumors were collected at Fuzhou Sheng-Li Hospital (2 M/2 F, ages: 46-67). Surgical specimens were digested to single cells, seeded into matrigel, and cultured with GC PDO medium (Biogenous). PDOs (200/well) were treated with 0 – 10 μM DCLK1-IN-1 for 3 days, and counted and imaged daily. Finally, PDOs were stained with calcein-AM and PI and subjected to CellTiterGlo viability assay. GC cells and PDOs were treated with 0 – 10 μM DCLK1-IN-1 for 24 h for Western blot analysis. Furthermore, PDOs were processed for immunohistochemistry of DCLK1. Statistical analyses were performed in GraphPad Prism.
Results
Functional assays for DCLK1-IN-1 in GC cell lines revealed dose-dependent inhibition of colony and spheroid formation. Furthermore, DCLK1-IN-1 downregulated CSC markers LGR5, CD133, and C-Myc. To confirm these findings, we overexpressed DCLK1-WT and DCLK1-D533N (kinase-dead), and found that DCLK1-D533N mimicked DCLK1-IN-1 treatment molecularly and functionally.
IHC staining showed that DCLK1 was specifically expressed in GC PDOs. After DCLK1-IN-1 intervention, significant inhibition of PDO number and viability was revealed.
Conclusion
These results suggest that DCLK1 kinase inhibition can attenuate GC stemness and tumorigenesis in patient-relevant models, and support further development of DCLK1 targeted therapies. Future work should focus on unraveling the function of DCLK1 isoforms to gain a more specific therapeutic understanding of DCLK1 in GC.