Identification and Early Intervention in Key Subpopulations of Pancreatic Cancer Liver Metastases
Authors
Wei Wu1,2#, Miao Yu2#, Qianghu Wang1, 2, 3, 4, 5*
Affiliations
- School of Biological Science & Medical Engineering, Southeast University, Nanjing, Jiangsu 210000, China
- Department of Bioinformatics, Nanjing Medical University, Nanjing, Jiangsu 211166, China
- Collaborative Innovation Center for Personalized Cancer Medicine, Nanjing Medical University, Nanjing, Jiangsu 211166, China
- The Affiliated Cancer Hospital of Nanjing Medical University, Jiangsu Cancer Hospital, Jiangsu Institute of Cancer Research, 210002 Nanjing, China.
- The First Affiliated Hospital of Nanjing Medical University, Nanjing 210029, China
Introduction
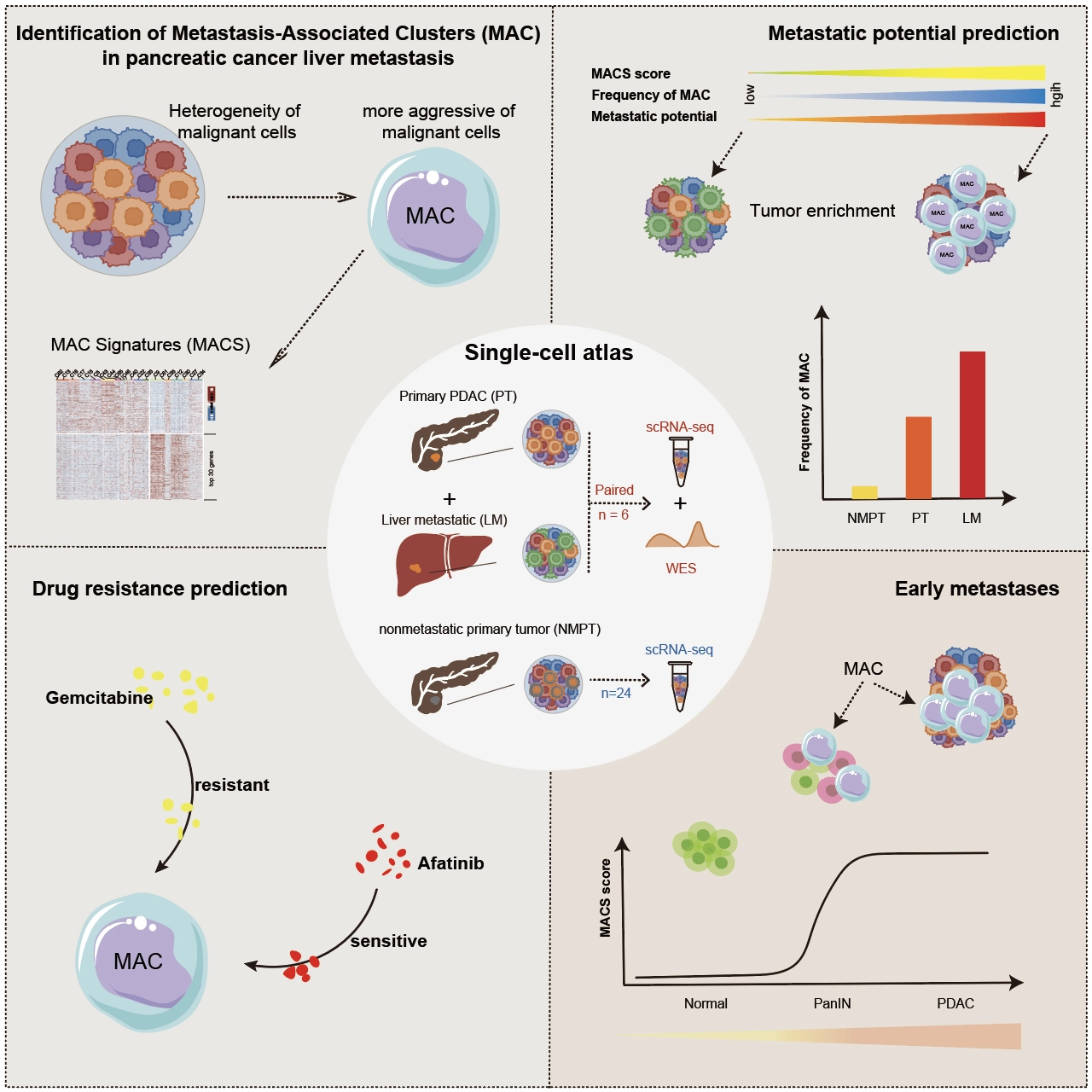
Metastatic pancreatic cancer has a dismal 5-year survival rate of only 3%, largely due to the absence of early detection and effective treatments. Previous research indicates that only specific subpopulations of tumor cells are capable of distant metastasis, yet their precise identification and profiling remain elusive. This study aims to identify these dominant subgroups in pancreatic cancer liver metastasis, determine their emergence timing, and explore their therapeutic potential.
Methods
Employing single-cell and spatial transcriptome analyses on human pancreatic ductal adenocarcinoma (PDAC) liver metastases, we developed a computational method to characterize the Metastatic-Associated Clusters Signature (MACS). This algorithm analyzed multiple omics databases, with findings validated through clinical data, immunohistochemistry, and cellular experiments to ascertain the roles of specific metastatic subpopulations and identify therapeutic targets.
Results
This study constructed a single-cell atlas of paired samples from primary pancreatic cancer and liver metastases, identifying and verifying metastatic-associated clusters (MAC) and their molecular markers. Through the integration of clinical retrospective data, we discovered that the molecular characteristics of MAC could serve as potential markers for assessing the risk of PDAC metastasis. These markers are highly expressed at the PanIN stage, indicating that MAC manifests early in tumor progression. Further analysis revealed that MAC is resistant to gemcitabine treatment yet sensitive to afatinib. LMO7 was identified as a key molecule mediating resistance to gemcitabine; its knockdown enhanced sensitivity to this treatment. Conversely, afatinib effectively inhibited the metastasis of tumor cells overexpressing LMO7.
Conclusion
The discovery of MAC provides new insights into the molecular heterogeneity of metastatic pancreatic cancer and suggests that MAC characteristics can be used as potential molecular markers for predicting tumor metastasis, drug sensitivity, and guiding combination therapy. This research offers novel ideas and potential clinical value for personalized diagnosis and treatment in patients.