Pathological Complete Response (PCR) to 5-Fluorouracil, Leucovorin, Oxaliplatin and Docetaxel (FLOT) ± Durvalumab (D) in Resectable Gastric and Gastroesophageal Junction Cancer (GC/GEJC): Subgroup Analysis by Region from the Phase 3 MATTERHORN Study
Authors
Yelena Y. Janjigian1, Salah-Eddin Al-Batran2, Zev A. Wainberg3, Eric Van Cutsem4, Daniela Molena5, Kei Muro6, Woo Jin Hyung7, Lucjan Wyrwicz8, Do-Youn Oh9, Takeshi Omori10, Markus Moehler11, Marcelo Garrido12, Sulene C.S. Oliveira13, Moishe Liberman14, Victor Castro Oliden15, Mehmet Bilici16, John F. Kurland17, Ioannis Xynos18, Helen Mann18, Josep Tabernero19
Affiliations
- Gastrointestinal Oncology Service, Memorial Sloan Kettering Cancer Center, New York, NY, USA;
- Institute of Clinical Cancer Research, Krankenhaus Nordwest, University Cancer Center, Frankfurt, Germany;
- Department of Gastrointestinal Medical Oncology, David Geffen School of Medicine at UCLA, Los Angeles, CA, USA;
- Department of Gastroenterology/Digestive Oncology, University Hospitals Leuven and KU Leuven, Leuven, Belgium;
- Division of Thoracic Surgery, Memorial Sloan Kettering Cancer Center, New York, NY, USA;
- Department of Clinical Oncology, Aichi Cancer Center Hospital, Nagoya, Japan;
- Department of Surgery, Yonsei University College of Medicine, Seoul, Republic of Korea;
- Department of Oncology and Radiotherapy, Maria Sklodowska-Curie National Research Institute of Oncology, Warsaw, Poland;
- Division of Medical Oncology, Department of Internal Medicine, Seoul National University Hospital; Cancer Research Institute, Seoul National University College of Medicine, Seoul, Republic of Korea;
- Department of Gastroenterological Surgery, Osaka International Cancer Institute, Osaka, Japan;
- Research Center for Immunotherapy (FZI), Johannes Gutenberg-University Clinic, Mainz, Germany;
- Hemato-Oncology Department, SAGA Clinical Trial Centre and Universidad Mayor, Santiago, Chile;
- Clinical Oncology, The Clinical Research Center, Northern Riograndense League Against Cancer, Natal, Rio Grande do Norte, Brazil;
- Division of Thoracic Surgery, Department of Surgery, Centre Hospitalier de l'Université de Montréal, Centre de Recherche du CHUM, Montréal, Quebec, Canada;
- National Institute of Neoplastic Diseases (INEN), Lima, Peru;
- Department of Medical Oncology, Atatürk University Faculty of Medicine, Erzurum, Turkey;
- AstraZeneca, Gaithersburg, MD, USA;
- AstraZeneca, Cambridge, UK;
- Medical Oncology Department, Vall d’Hebron Hospital Campus & Institute of Oncology (VHIO), IOB-Quiron, UVic-UCC, Barcelona, Spain
Introduction
FLOT was established as a perioperative therapy for GC/GEJC following the Phase 2/3 FLOT4 study conducted in Germany (Al-Batran et al, Lancet Oncol 2016). MATTERHORN (NCT04592913) showed a significant improvement in pCR with perioperative D + FLOT vs placebo (P) + FLOT in GC/GEJC at first interim analysis (Janjigian et al, ESMO Congress 2023). Subgroup analyses by region and country were completed to assess pCR rates with FLOT and benefit of D + FLOT across the global study population.
Methods
Participants (pts) with resectable GC/GEJC were randomized 1:1 to D 1500 mg or P every 4 weeks (Q4W) on Day 1 plus FLOT Q2W on Days 1 and 15 for 4 cycles (2 doses of D or P and 4 doses of FLOT pre- and post-operative), followed by D 1500 mg or P on Day 1 Q4W for 10 cycles. Randomization was stratified by Asia vs non-Asia. pCR (Modified Ryan; central review) was assessed in prespecified (Asia) and post-hoc regional subgroups, including 6 countries with the highest numbers of randomized pts.
Results
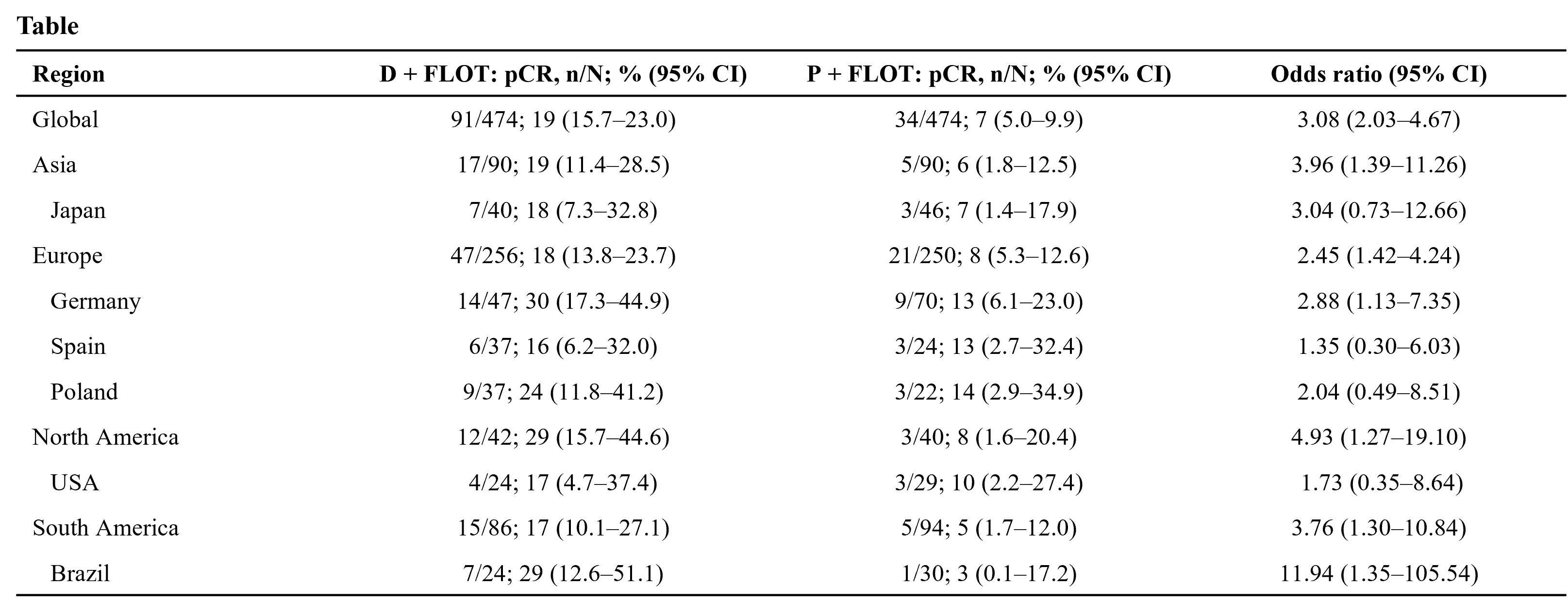
Of 948 pts randomized globally, 180 pts (19%) were in Asia. pCR outcomes with FLOT in Asia were consistent with global outcomes. pCR rates were improved with D + FLOT vs P + FLOT in all regions (Table), despite some imbalances in baseline characteristics and numerical differences in pCR rates by geographic location. pCR rate with P + FLOT in the German subgroup (13%) was similar to FLOT in the FLOT4 study (16%). pCR with D + FLOT vs P + FLOT was improved across country subgroups. Similar trends across regional subgroups were observed for combined complete and near-complete response rate.
Conclusion
In MATTERHORN, pCR was consistently improved with perioperative D + FLOT across geographic regions. The study is ongoing for the primary objective of event-free survival.